In recent years, there have been developed several new treatments for rheumatic disease, which can be more effective in controlling the treatment and improving patients’ living quality. However, it might also lead to a decrease in immunity and raise the risk of hepatitis B reactivation.
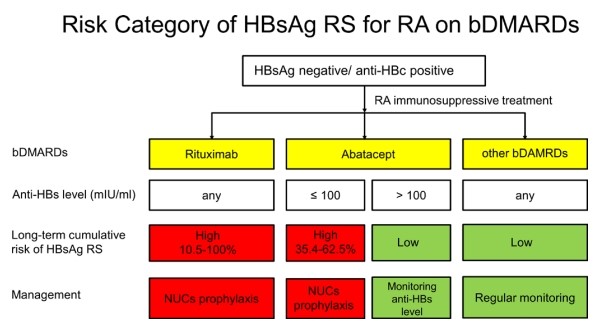
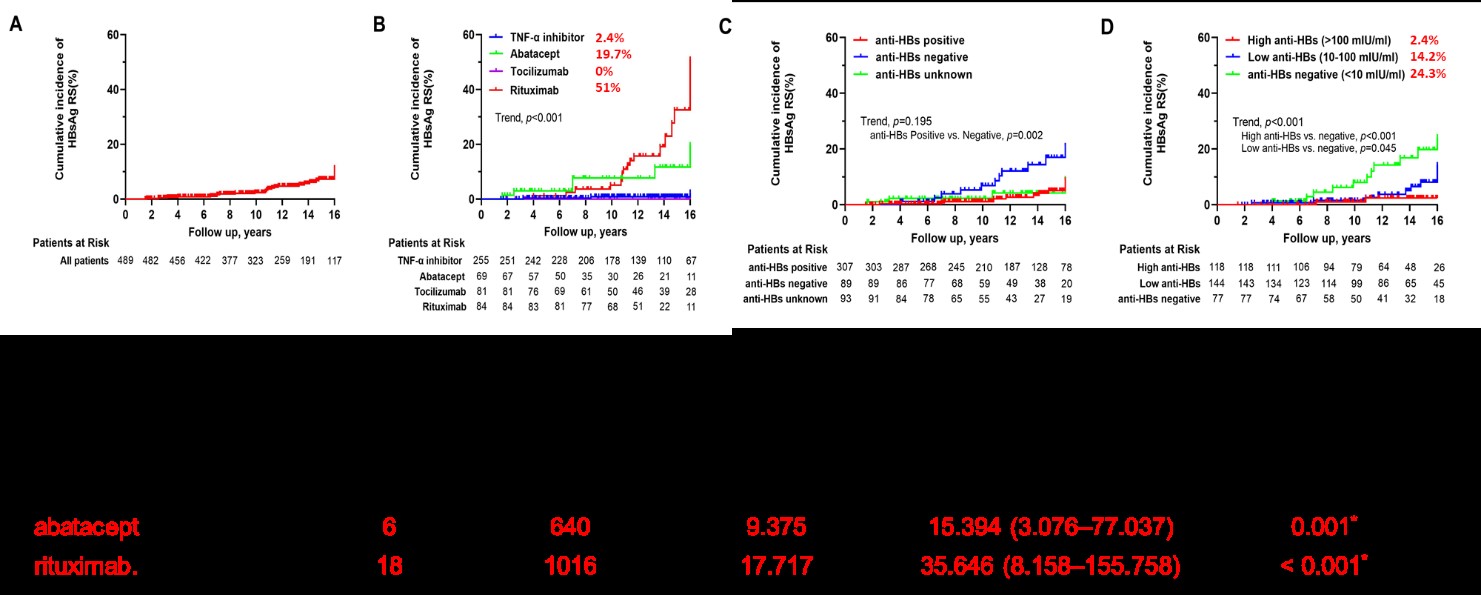
The research is the first report of discovering that abatacept (CTLA-4 fusing Ig) is second to rituximab at risk of HBsAg reverse seroconversion. The research objects are 1,937 rheumatoid arthritis patients who receive all kinds of Immunosuppressive drug treatments. After analyzing long-term tracking results, abatacept is discovered to be second to rituximab at risk of HBsAg reverse. The research has also indicated that patients who already have anti-HBs within their blood flow might still lose their antibodies and the HBsAg might still be reactivated throughout the treatment. As a result, it should be classified as high-risk of HBV if anti-HBs levels are not high enough, at which, antiviral prophylaxis is highly recommended.
More information: https://www.nycu.edu.tw/news/2741/




